Optogenetic Sensitivity Relief explores light-based modulation of nerve responses. Could this innovation qualify for Breakthrough Device Designation? This blog examines feasibility, benefits, and regulatory considerations. It highlights how emerging technologies may accelerate approval pathways.
Optogenetic Sensitivity Relief uses light to influence nerve activity. It targets sensitivity pathways with high precision. This approach may reduce pain without traditional chemical agents.
Breakthrough Device Designation is granted by regulatory authorities for innovative technologies. It applies to devices addressing unmet medical needs. This pathway enables faster development and review processes.
Optogenetic Sensitivity Relief offers targeted and minimally invasive treatment options. It may improve patient outcomes in sensitivity management. This aligns with criteria for Breakthrough Device Designation consideration.
Regulatory approval requires strong clinical evidence. Optogenetic Sensitivitymust prove consistent and safe outcomes. Data must support eligibility for Breakthrough Device Designation.
Complex integration of optics and biology creates challenges. Optogenetic Sensitivity requires precise calibration and validation. Meeting standards is critical for Breakthrough Device Designation success.
Achieving Breakthrough Device Designation offers competitive benefits. It accelerates time to market and enhances credibility. Optogenetic Sensitivity can position brands as innovation leaders.
Optogenetic Sensitivity shows potential for advanced sensitivity treatment. Its alignment with innovation criteria supports Breakthrough Device Designation consideration. For B2B manufacturers, this pathway offers strategic growth opportunities. Contact us

.jpg)
Can Cartoons Improve Brushing Habits?

The Top Things You Need to Know About Teeth Whitening
.jpg)
Pump Failure Plus Weak Suction – Terminal Malfunction?

Electric toothbrushes: personalized oral care solutions

How to choose a reliable electric toothbrush factory?

Need a Toothbrush Factory Price for Bulk Toothbrush Orders?

How Important Is Waterproof Testing for Electric Toothbrushes?

How Does Jet Stream Technology Function Efficiently in a Simple Gravity Feed System?
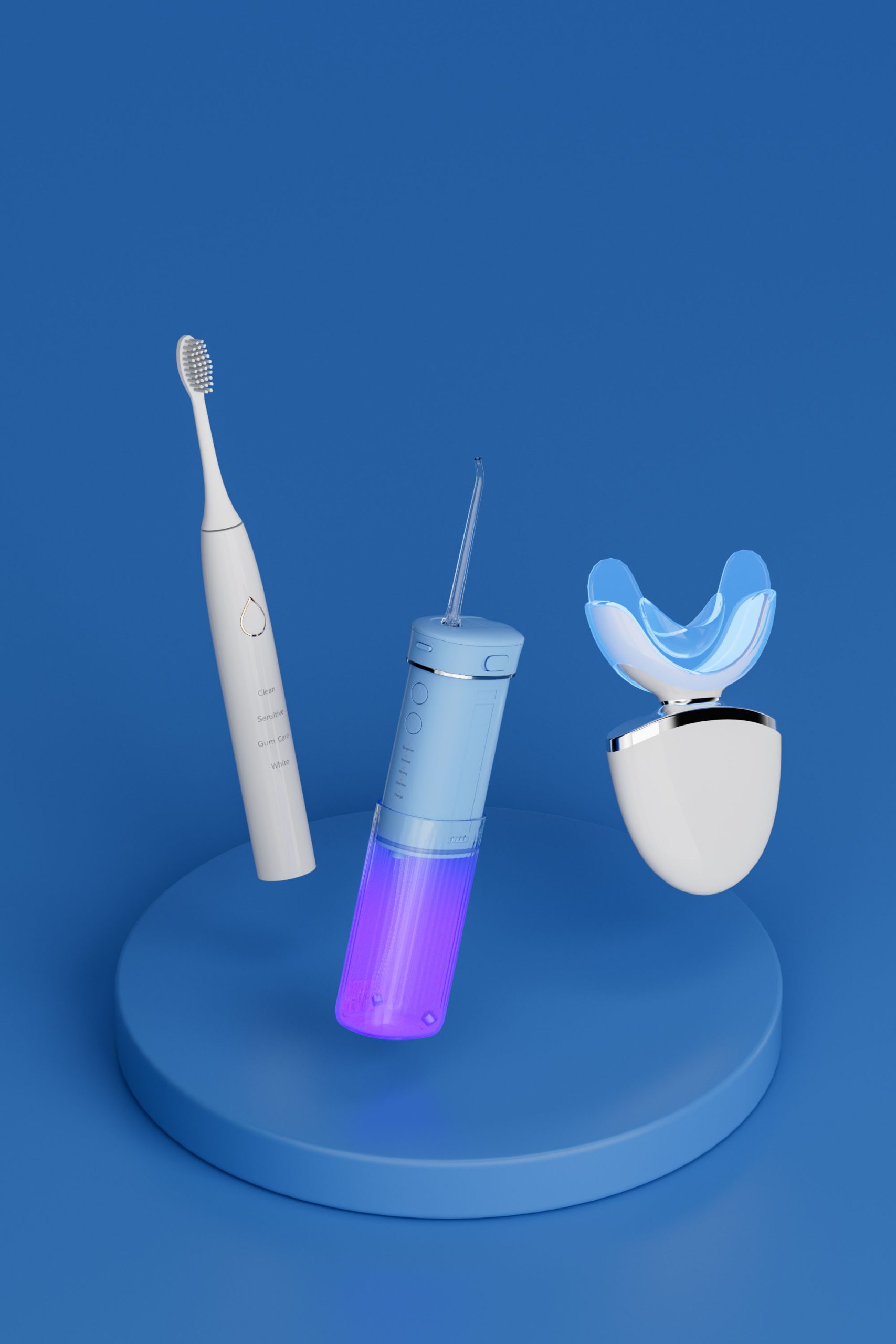
Latest Trends in Waterproof Technology for Electric Toothbrush Future Directions and Application Scenarios

Oral Care in Cosmetics: The Next Frontier for Beauty Brands & OEMs
.jpg)
Morning vs. Night Brushing Routine
.jpg)
Smart Electric Toothbrush Manufacturer

Clean & Maintenance Tips for Your Sonic Electric Toothbrush

What is Teeth Whitening Precare and Aftercare?
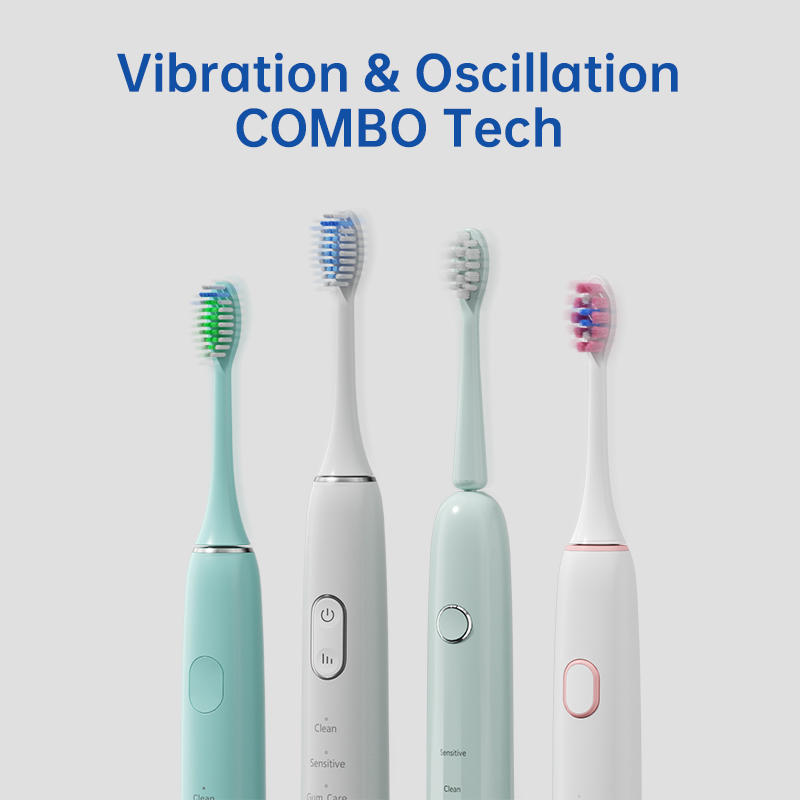
Travel Electric Toothbrush OEM: Compact and Portable Designs
Best Dentist-Recommended Braces Cleaning Products for Braces Wearers