Launching your own whitening kit brand can be an exciting and profitable venture — but navigating the complex world of regulations is often less glamorous. Whether you’re an established oral care brand expanding your product line or a startup exploring OEM partnerships, understanding the OEM Whitening Kits regulations is essential for success. Below, we break down how to simplify OEM market access, avoid regulatory pitfalls, and build a foundation of compliance expertise that protects your brand and customers.
Before you launch your whitening kit, it’s vital to understand how regulations differ by region.
For example, the European Union classifies teeth whitening products containing certain levels of hydrogen peroxide as cosmetic or medical devices, depending on their concentration. In the U.S., the FDA has specific labeling and safety requirements, while in Asia, whitening agents are strictly monitored for consumer safety.
Working closely with your OEM manufacturer ensures that your formulation, packaging, and labeling comply with local whitening kit regulations. A knowledgeable factory partner can help you prepare all the necessary documentation and certifications before your products hit the shelves.
The right manufacturing partner does more than just produce your OEM Whitening Kits — they pave the way for smooth OEM market access.
When evaluating potential factories, consider their regulatory history, testing capabilities, and experience with export markets. A reliable OEM should provide transparent quality control reports, third-party testing results, and compliance documentation to support your market entry strategy.
By collaborating with an OEM that already understands the target country’s regulatory expectations, you can significantly shorten your time to market and reduce costly delays.
Compliance isn’t something to think about after your product is ready. It should be integrated into your development process from day one.
Brands that invest in compliance expertise — whether through internal specialists or partnerships with experienced OEM manufacturers — can design products that are market-ready from the start. This proactive approach ensures that everything from ingredient selection to product labeling meets legal standards and passes safety evaluations.
Remember: a well-documented compliance process not only protects you legally but also builds consumer trust and brand credibility.
Regulatory non-compliance isn’t just a legal issue — it’s a brand reputation risk. To ensure risk avoidance, work with manufacturers who follow international quality management systems like ISO 22716 (cosmetics GMP) or ISO 13485 (medical devices).
Implementing regular audits, product testing, and traceability systems allows you to identify and correct issues before they become regulatory violations. Prevention is always less costly than correction.
Your product documentation and labeling often determine whether your whitening kit gains or loses market access. Ensure your OEM partner can assist with:
Ingredient safety data sheets
Certificates of analysis
Product Information Files (PIFs)
Label translations and claims verification
Accurate documentation is the backbone of regulatory approval and helps avoid unnecessary delays during import or inspection.
In a crowded oral care market, compliance can become your differentiator.
By demonstrating that your whitening kits meet international standards, you signal to distributors and customers that your brand is trustworthy and high-quality.
Partnering with an OEM that offers strong compliance expertise and understands the nuances of whitening kit regulations empowers your business to expand globally — with confidence and risk avoidance built into every step.
Navigating the maze of whitening kit regulations doesn’t have to be overwhelming. With the right OEM partner, clear documentation, and a proactive compliance mindset, market access becomes simple and strategic.
For oral care brands ready to scale, mastering regulatory compliance is not just about following the rules — it’s about unlocking new markets and long-term growth.
.jpg)
User Adaptation Struggles from Improper Angle Use? Device Design Matters!

When Selecting Products for the US Market, What Kind of Aesthetic Electric Toothbrushes Are Most Popular?
.jpg)
The Potential of the Oral Care Market in Southeast Asia: Trends & Opportunities for Brands

How Does a UV Sanitizing Component Complement the Use of Food-grade Bristle Material?
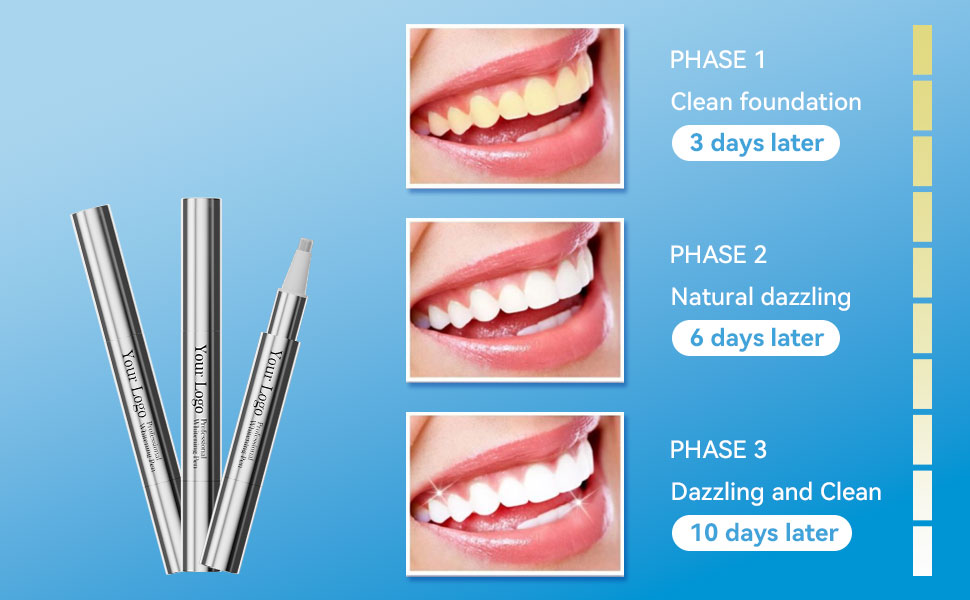
A Comprehensive Guide to Teeth Whitening Gel Ingredients

Can a Wireless Whitening Kit Integrate Foam from a Teeth Whitening Foam Manufacturer for Innovation?
.jpg)
Top 5 Factors When Selecting a UV Sterilizing Water Flosser Manufacturer

Sonic vs Rotary: Which Electric Toothbrush Technology is Better for Your Brand?
.jpg)
Professional Dental Care Toothbrush Bulk | Trusted Hygiene for Clinics
.jpg)
Could Bristle Splaying Rapidly Induce Uneven Cleaning Patterns?
.jpg)
Red Light Gum Therapy Whitening Device Supplier | LED Oral-Care OEM Manufacturer
Can an ADA Accepted Water Flosser Be Compactly Designed by a Travel Water Flosser Manufacturer?

A Comprehensive Analysis of the Pros and Cons of Water Flossers: Product Optimization from water flosser Manufacturers’ Perspective
.jpg)
Blue Light Electric Toothbrush OEM Manufacturing Solutions
.jpg)
Can Sweeping Oscillation Technology Be Integrated into Your OEM Manufacturing Line?

How Do Quality Control Protocols Contribute to Rigorous Quality Assurance?