As demand grows for fast and visible whitening results, an Accelerated Whitening Solution must balance performance with safety and legality. For manufacturers and OEM brands, achieving speed alone is not enough—full Aesthetic Device Compliance is essential to ensure market access, risk control, and long-term brand credibility in professional and consumer channels.
An Accelerated Whitening Solution often falls under aesthetic or cosmetic device regulations. Aesthetic Device Compliance requires clear classification, documented risk assessment, and alignment with applicable regional standards.
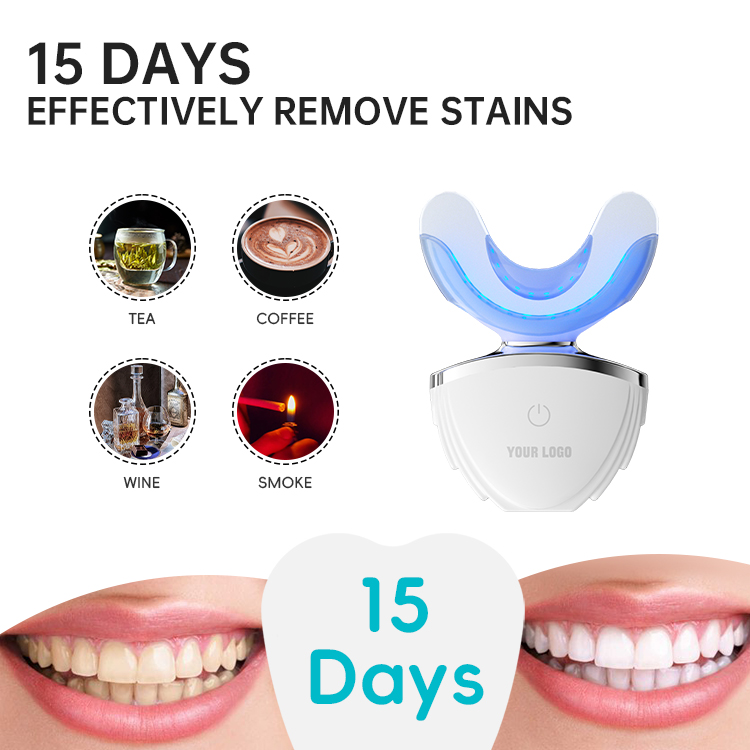
Accelerated whitening systems typically combine gels, light sources, or heat. Demonstrating Aesthetic Device Compliance means validating how these elements interact safely within the Accelerated Whitening Solution under real-use conditions.
Components that contact the mouth must meet strict material standards. Full Aesthetic Device Compliance ensures that materials used in an Accelerated Whitening Solution are biocompatible, non-toxic, and suitable for repeated exposure.
Timers, output limits, and auto-shutdown functions are critical. These safety features support Aesthetic Device Compliance by preventing misuse and ensuring controlled operation of the Accelerated Whitening Solution.
Regulators and B2B buyers expect transparent documentation. A compliant Accelerated Whitening includes test reports, technical files, and traceability records that demonstrate adherence to Aesthetic Device Compliance requirements.
Lack of compliance can result in recalls, delays, or bans. Full Aesthetic Device Compliance safeguards the commercial viability of an Accelerated Whitening and strengthens trust with distributors, clinics, and OEM partners.
An Accelerated Whitening is only truly market-ready when it is backed by full Aesthetic Device Compliance. For B2B manufacturers, compliance is not a barrier to innovation—it is a foundation that ensures safety, scalability, and sustainable growth across global markets. Contact us


Is the Specific LED Wavelength the Key to Being Truly Enamel Safe?

Can a Whitening Device Manufacturer Develop an Integrated At-Home Whitening System?
.jpg)
Dual-Color Light Therapy Toothbrush OEM – LED Oral-Care Innovation

LED Teeth Whitening Kits: Maximizing Value with the Right OEM Partner

Why Should a Cosmetic Grade Whitening Gel Come with a Precision Gel Applicator?
.jpg)
multi-mode electric toothbrush supplier

Why Are Safe Whitening Ingredients Paramount in a Wearable Whitening Strip?
.jpg)
Graduation Gift Sonic Toothbrush Houston
.jpg)
Bulk sonic toothbrushes for businesses Houston
.jpg)
sonic toothbrush with smart pressure sensor

Can a Torque Control Motor Benefit from Advanced Kinematic Pair Design in Electric Toothbrushes?
.jpg)
Electric Toothbrush with Competitive Features for Global Buyers
.jpg)
Powsmart-bulk oral care Houston

How Does a Whitening Pen Embody the Ultimate Portable Design for On-the-Go Care?
How Is Whitening Device Safety Engineered Into a Portable Whitening Kit?

How Does a Teeth Whitening Supplier Ensure Whitening Kit Certification for Market Safety?