In the oral care manufacturing industry, achieving Brush Head Compatibility across multiple devices is often seen as a commercial advantage. However, from a regulatory perspective, universal designs introduce significant complexity in meeting Certification Compliance requirements. For OEMs and brand owners, balancing modular compatibility with strict safety, performance, and regional certification standards remains a critical challenge.
Universal brush heads must fit multiple handle designs, which can lead to inconsistent locking mechanisms or tolerances. Certification Compliance often requires validated retention force and detachment safety for each configuration, complicating approval processes.
Brush Head Compatibility becomes more complex when devices use different drive systems, vibration frequencies, or power outputs. Certification bodies may require separate testing to confirm safe operation under each electrical or mechanical interface.
Universal brush heads are exposed to varied operating conditions. Materials must meet Certification Compliance standards for wear, chemical resistance, and biocompatibility across all compatible devices, increasing testing scope and documentation.
Regulatory frameworks often require consistent cleaning efficacy and user safety. Ensuring Brush Head Compatibility does not compromise performance across different platforms can trigger additional validation and repeat testing.
Global markets apply different certification standards (FDA, CE, ISO). A universally compatible brush head may meet requirements in one region but fail Certification Compliance criteria in another, limiting true global standardization.
Universal designs complicate traceability. Any design change to improve Brush Head Compatibility may require re-certification across multiple devices, increasing time-to-market and compliance costs.
While universal Brush Head Compatibility offers supply chain and consumer benefits, it significantly increases the complexity of Certification Compliance. For B2B manufacturers and OEM partners, a strategic approach—combining modular design, early regulatory alignment, and rigorous validation planning—is essential to achieving scalable, compliant oral care solutions. Contact us


How Does Packaging Customization Service Assist with Aesthetic Device Compliance?
.jpg)
How Does Desktop Irrigator Design Integrate an Anti-bacterial Hose for Long-term Hygiene?

How Does Jet Stream Technology Function Efficiently in a Simple Gravity Feed System?
.jpg)
Dual-Color Light Therapy Toothbrush OEM – LED Oral-Care Innovation

How Does Including a Wireless Charging Module Influence Kit Packaging Design?
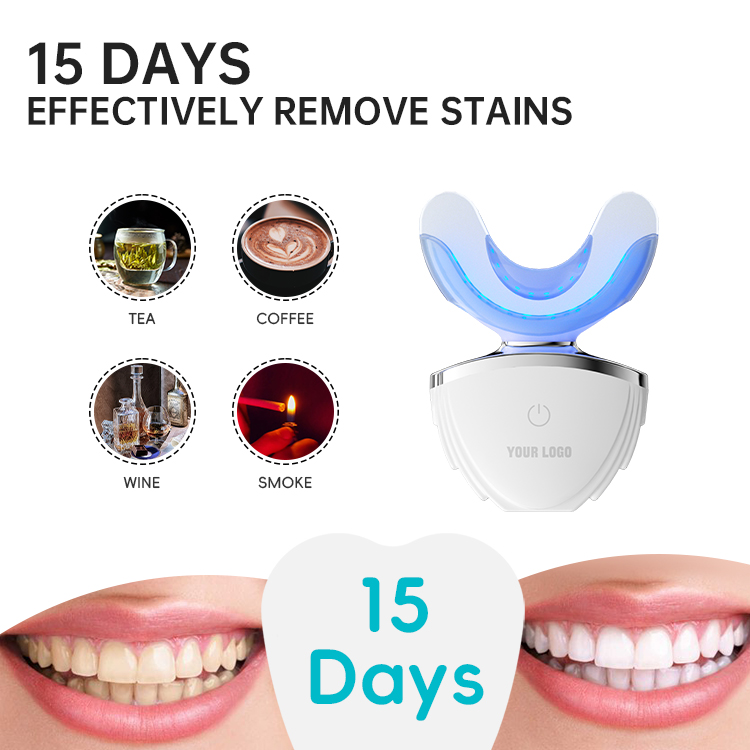
How Does an LED Whitening Lamp Activate a Specific Whitening Gel Formulation Effectively?
.jpg)
sonic toothbrush with smart pressure sensor
.jpg)
Whitening Device Packaging Customization for OEM and ODM Brands

How Does a Whitening Pen Embody the Ultimate Portable Design for On-the-Go Care?
.jpg)
Ergonomic Sonic Toothbrush Handle OEM
.jpg)
Graduation Gift Sonic Toothbrush Houston

How Does a Linear Resonant Actuator Benefit from an IPX8 Waterproof Solution in Electric Toothbrushes?
Professional Sonic Toothbrush Factory in Houston, Texas

Should Nozzle Material Always Be Medical-grade Material for Safety?

How Does Accelerometer-based Feedback Integrate With a Pressure-sensitive Matrix for Smart Brushing?
.jpg)
Cordless Water Flosser for Family Use – Portable Home Oral Hygiene Solutions