Toothbrush export documentation is a core requirement for OEM manufacturers shipping products to overseas markets. Missing or incorrect paperwork often leads to customs delays, additional costs, or even shipment rejection.
For B2B buyers, understanding document requirements improves supply chain reliability and planning accuracy.
These documents declare product value, HS codes, quantities, and shipment details. Accuracy is essential to avoid customs disputes.
Depending on destination, a COO may be required to qualify for preferential tariffs or regulatory clearance.
Electric toothbrushes usually require CE, FCC, or similar conformity declarations. These documents confirm that products meet safety and electromagnetic standards.
Export requirements vary across regions. EU markets emphasize CE compliance and technical files, while U.S. imports focus on FCC documentation and labeling accuracy.
Therefore, toothbrush export documentation should always be prepared according to destination country rules rather than a one-size-fits-all approach.
Experienced OEM factories assist buyers by preparing standardized files, test reports, and shipment declarations. This support reduces buyer workload and minimizes clearance risks.
At Powsmart, export documentation is managed together with logistics planning to ensure shipment readiness. Relevant product references are available at:
👉 https://www.powsmart.com/sonic-electric-toothbrush/
Incorrect HS codes, mismatched product descriptions, and outdated certificates are frequent issues. Moreover, inconsistent labeling between packaging and invoices may trigger inspections.
By reviewing all files before shipment, buyers can significantly reduce these risks.
For international trade compliance and customs guidance, buyers may consult World Customs Organization (WCO) publications.
Standardizing document templates and working with experienced partners simplifies repeated shipments. Over time, this improves efficiency and reduces unexpected delays.
Toothbrush export documentation is therefore not just paperwork, but a key component of reliable global supply chains.
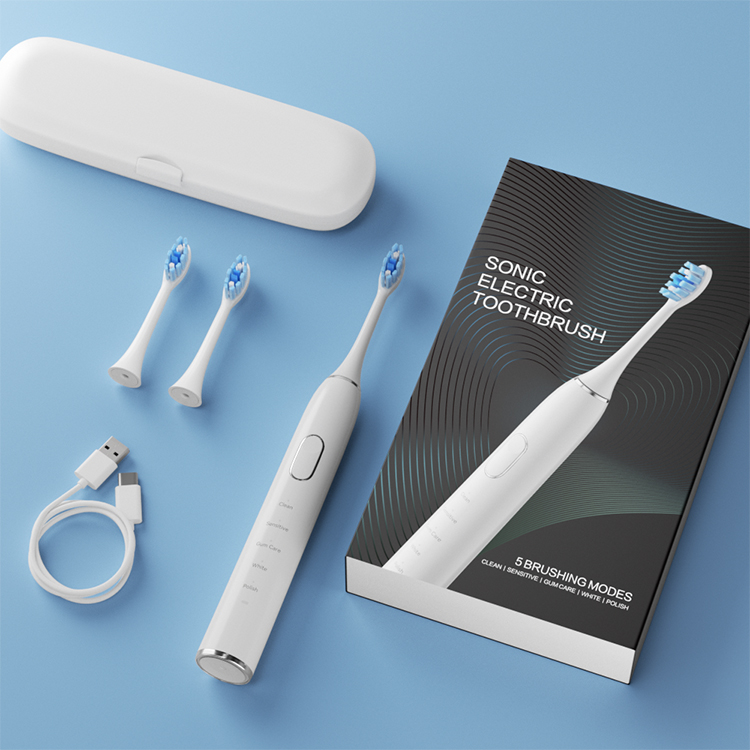
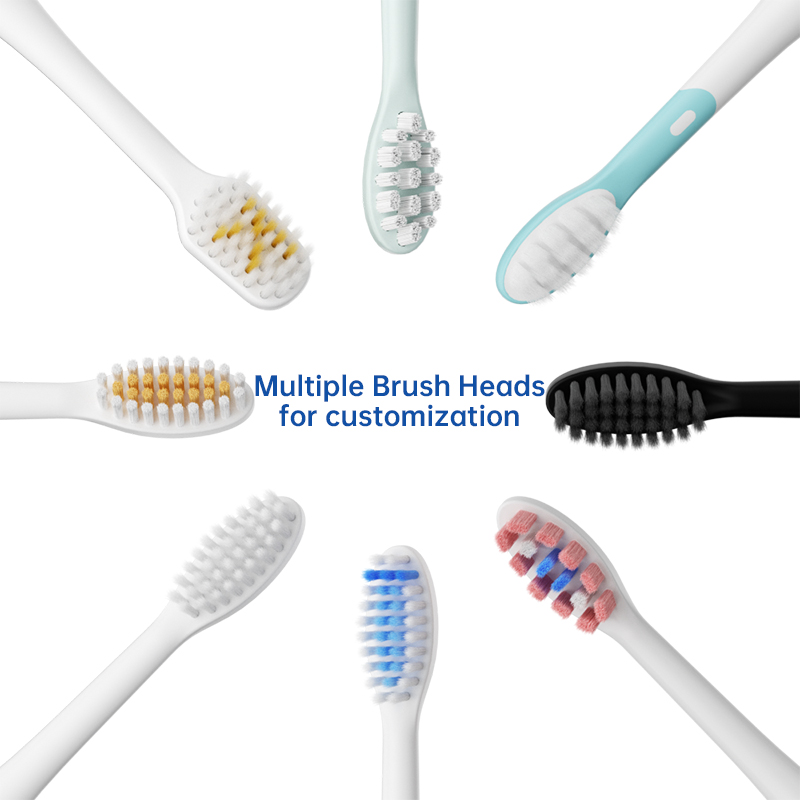
How Does Integrated Brush Head Sterilization Add Value to Travel Case Manufacturing?

Does the Efficiency of a Motor Controller IC Directly Impact Battery Life Optimization?
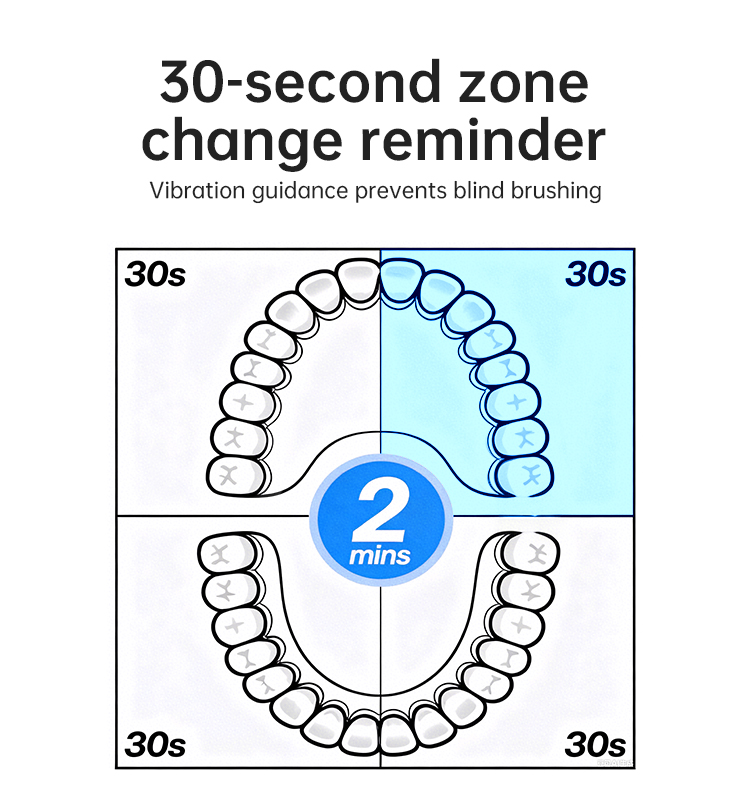
Is a High-Pressure Water Pump Effective Without a Robust Leak-Proof Valve?
.jpg)
Best Electric Toothbrush for Kids in Chicago
.jpg)
USB rechargeable sonic toothbrush supplier
.jpg)
Electric Toothbrush Sample Lead Time for OEM Projects
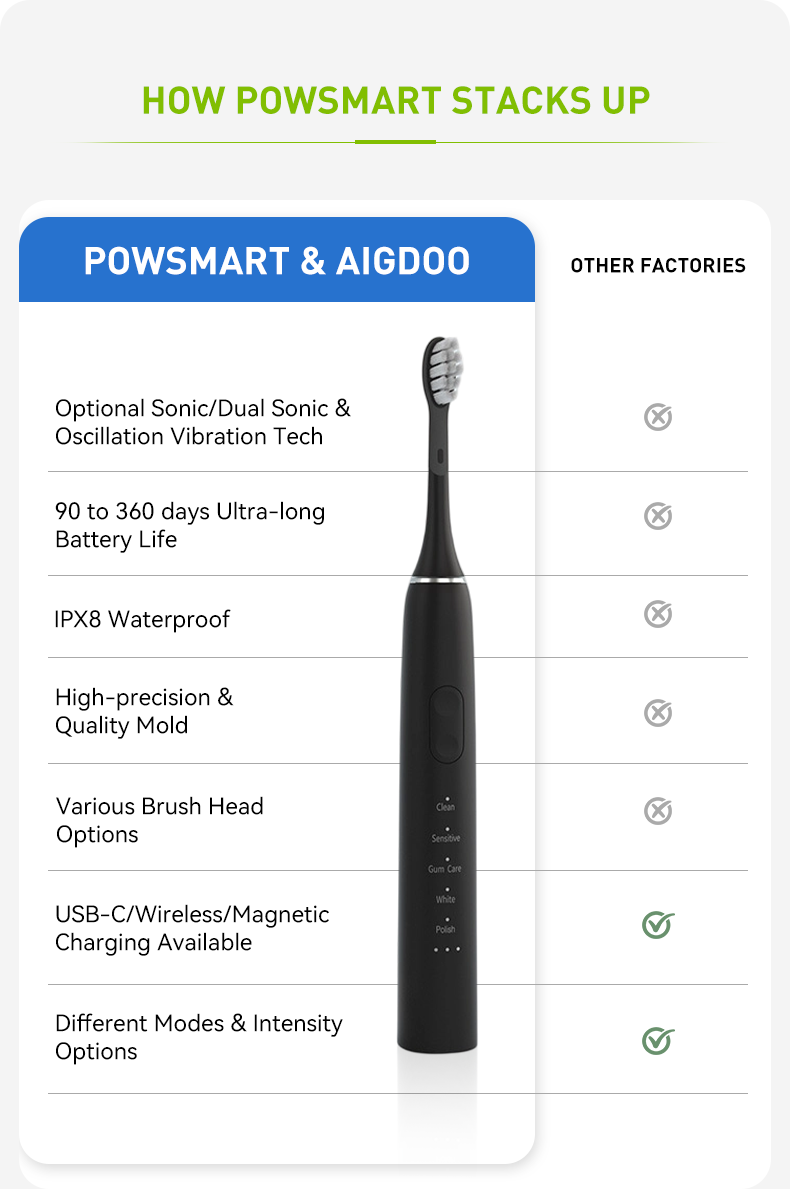
How Does Specialized Orthodontic Nozzle Design Affect the Requirements for Nozzle Storage?
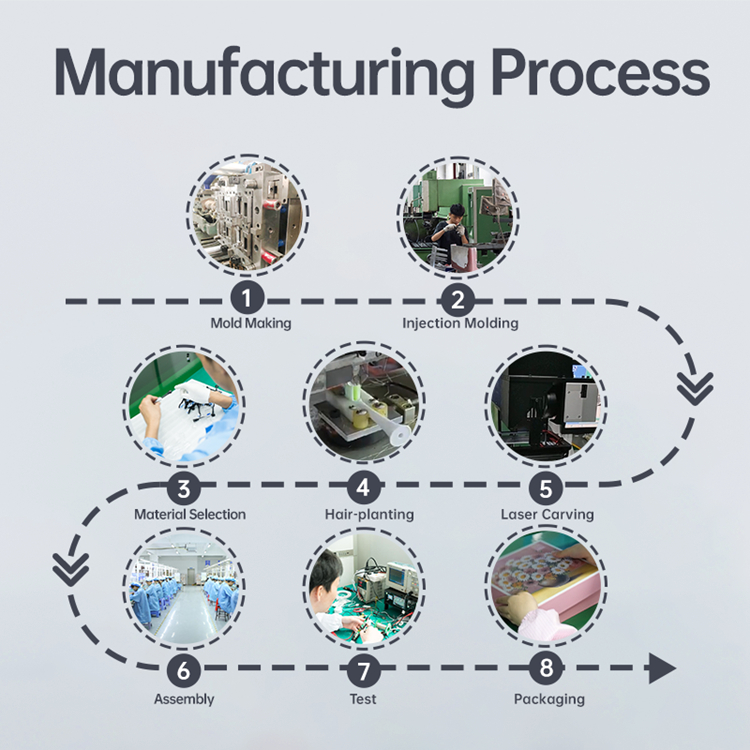
Looking for an OEM Partnership Program that Guarantees Long-Term Toothbrush Supply?
.jpg)
sonic electric toothbrush Chicago

Is Clear Instruction in the User Manual Vital for Proper Pressure Stabilization Use?
.jpg)
Which Electric Toothbrush is Best for Kids in Chicago?
-3-scaled.png)
Are You a Teeth Whitening Kit Manufacturer Serving the Beauty Salon Supply Industry?
.jpg)
Why Choose ADA Approved Electric Toothbrush in Chicago?
.jpg)
electric toothbrush with fast charging OEM

How Does Osmotic Pressure Regulation Fare in Accelerated Aging Tests for Product Longevity?

Can Sonic Wave Technology Benefit from a Motor Efficiency Upgrade?