For B2B product teams and OEM/ODM partners, the words Medical professional brush and Surgical grade toothbrush are sometimes used interchangeably — but they point to very different engineering, regulatory and commercial requirements. Below I unpack six clear dimensions so you can specify the right product for clinics, hospitals or retail-medical channels and avoid costly rework.
First and foremost, start with purpose. A Medical professional brush designed for routine clinical oral care: hygiene checks, patient home-care instruction, or chairside cleaning by dental staff. By contrast, a Surgical grade toothbrush is intended for peri-operative or high-acuity environments where minimising microbial loads (pre-op mouth prep, instrumentation suites) and sterility/traceability are essential.
Therefore:
Next, materials differ by risk and lifecycle. For a Medical professional brush you typically see durable, cleanable polymers and corrosion-resistant metals suitable for frequent disinfection. For a Surgical grade toothbrush materials and surface chemistry are chosen to be sterilizable and non-reactive:
In short, surgical grade equals higher material scrutiny and traceable certificates. Company web: https://www.powsmart.com/product/electric-toothbrush/
Crucially, the Surgical grade toothbrush must meet sterility workflows. That drives process and cost:
By contrast, a Medical professional brush may ship non-sterile with cleaning/disinfection instructions and a validated reprocessing SOP for clinic reuse.
Regulatory pathways separate the two categories:
Therefore, early engagement with regulatory and QA is mandatory when you intend to supply surgical grade items to hospitals.
Design choices follow the lifecycle model:
Commercial teams must model total cost of ownership (per patient use) and environmental impact when proposing either route.
Finally, what you prove drives adoption:
Hospitals and clinics will evaluate both specs and operational fit — not just “looks nice.”
If you’re deciding which product to build or certify, use this checklist:
Bottom line: a Medical professional brush engineered for clinical hygiene and frequent use under standard disinfection regimes; a Surgical grade toothbrush is a higher-risk product that must meet sterility, material and regulatory standards for use in sterile or peri-operative environments. Treat them as separate product families — not mere finish or branding variants — to avoid compliance gaps and ensure clinical adoption.
.jpg)
.jpg)

Energy saving and emission reduction measures and effectiveness of electric toothbrush factory
.jpg)
LED Light Electric Toothbrush Factory: OEM Manufacturing & Innovation

How does Soda Affect Your Teeth?
.jpg)
Electric Toothbrush Christmas Gift Texas
.jpg)
Premium Electric Toothbrush OEM Service for High-End Oral Care Brands

Can Waterproof IPX7 Design Protect a Hydro-dynamic Bearing in Humid Environments?
.jpg)
Orthodontic Water Flosser Supplier for Professional Oral Care Brands
.jpg)
Rechargeable Adult Toothbrush Supplier for Global B2B Buyers
Do Black Friday Deals Boost Electric Toothbrush Sales in Chicago?
.jpg)
Electric toothbrush with soft bristles wholesale

Frequency and Tips for Using Your At-Home Mouthpiece Recommended by Your Dentist
.jpg)
Downtown Chicago Electric Toothbrush Repair Services

comprehensive oral care solutions: Why Consumers Prefer Them
.jpg)
Blue vs Red Light for Teeth Whitening: Choosing the Right OEM Technology
.jpg)
Smoothie Teeth Stains Guide | Blueberry Drinks
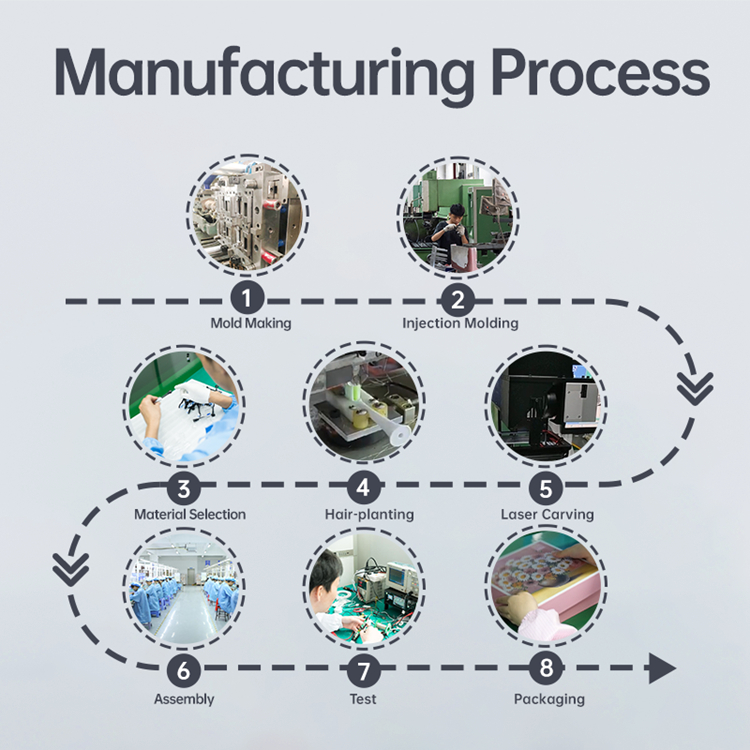
Sourcing Wireless Charging Coils? Need a Reliable Global Logistics Partner for Delivery?