For oral care devices and professional hygiene equipment, Pressure Stabilization is a critical function that directly affects safety, performance, and user confidence. However, even the most advanced technology can underperform if end users do not understand how to operate it correctly. This is why a clearly written and well-structured User Manual plays a vital role in ensuring proper use, especially in B2B-distributed products that serve diverse markets and user profiles.
Clear instructions in the User Manual guide users on how Pressure Stabilization works and when it is activated. Without proper explanation, users may apply incorrect pressure settings, leading to inconsistent results or potential device misuse.
Pressure Stabilization systems designed to adapt to different operating conditions. A detailed User Manual helps standardize user behavior, ensuring that the feature performs consistently whether the device is used in clinical, professional, or home environments.
When Pressure Stabilization limits clearly explained, users gain confidence in the device’s safety mechanisms. Transparent documentation reassures buyers and end users that the product engineered to protect both the device and the user during operation.
For B2B manufacturers, unclear documentation often results in higher customer service demand. A comprehensive User Manual reduces technical inquiries by clearly outlining setup, calibration, and pressure-related precautions, lowering long-term support costs.
In many markets, proper documentation is a regulatory requirement. Clearly defining Pressure Stabilization behavior within the User Manual helps demonstrate compliance with safety standards and supports smoother certification and audit processes.
A well-designed User Manual reflects the manufacturer’s commitment to quality. Clear explanations of advanced features like Pressure Stabilization enhance brand credibility and add value for OEM partners seeking reliable, professional-grade solutions.
Yes—clear instruction in the User Manual is vital for proper Pressure Stabilization use. For B2B manufacturers, effective documentation bridges the gap between engineering innovation and real-world application, ensuring safety, compliance, and long-term customer satisfaction while reinforcing brand trust in competitive markets. Contact us

.jpg)
Why choose a budget electric toothbrush from a trusted brand?

Deliver 7-Day Whitening: Partner with an OEM for Proven Oral Care Products
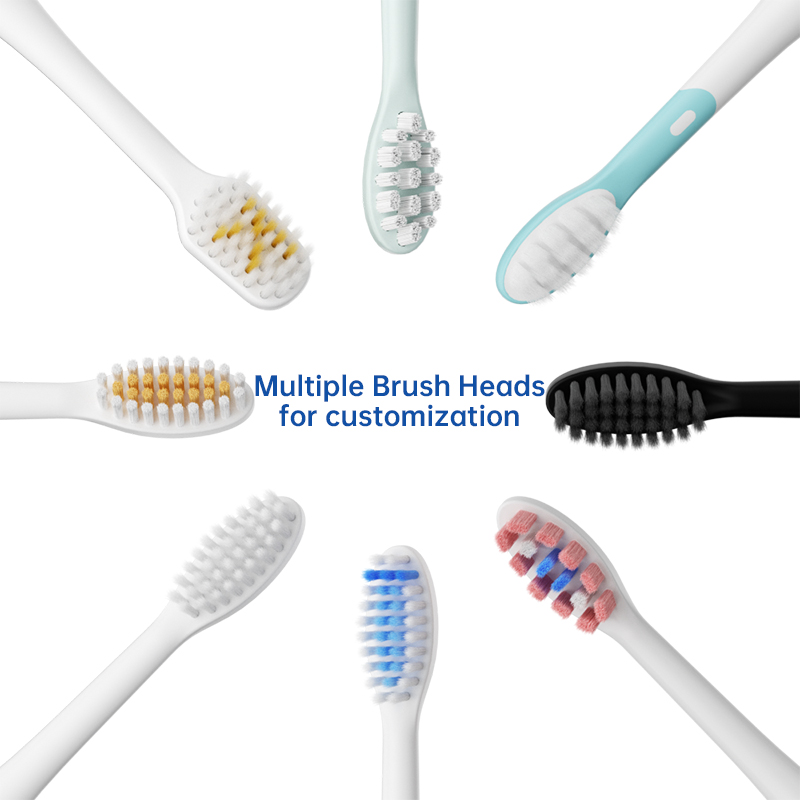
Extend Electric Toothbrush Head Life: An OEM Strategy for Customer Satisfaction
.jpg)
Oral Care Products Supplier in Houston TX

Why Are Safe Whitening Ingredients Paramount in a Wearable Whitening Strip?

How Does Integrated UV Sanitization Affect the Cartridge Replacement Cycle for Flossers?

How Can I Charge My UK Electric Toothbrush in the USA? Universal Charging Solutions from an OEM Factory
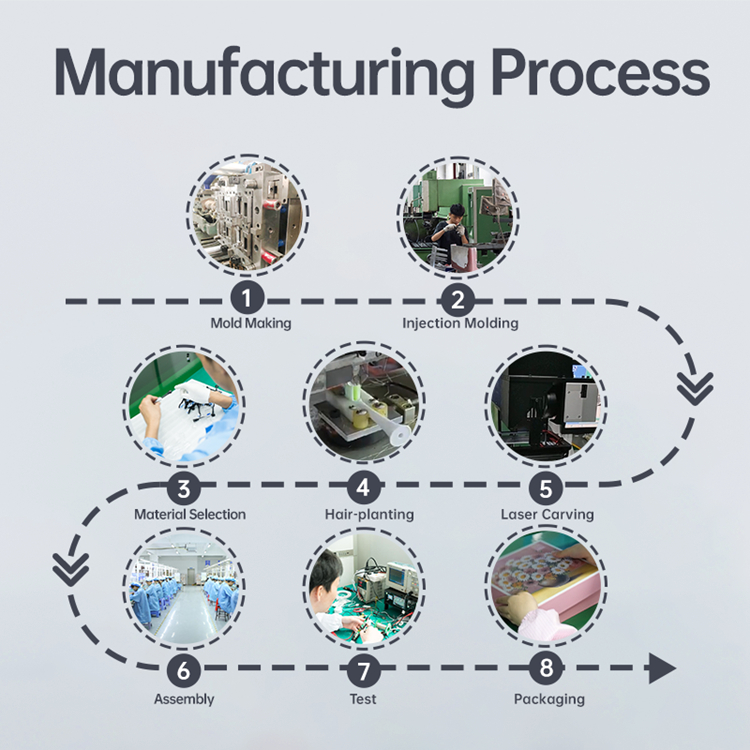
Sourcing Wireless Charging Coils? Need a Reliable Global Logistics Partner for Delivery?

Can Sonic Wave Technology Benefit from a Motor Efficiency Upgrade?
.jpg)
Private Label Electric Toothbrush Manufacturing with Low MOQ
.jpg)
Water Flosser for Braces Supplier for Orthodontic Care Markets

Multi-Packs and Family Sets – Electric Toothbrush OEM Packaging

Why Pair a Sonic Vibration Core with Food-grade Bristle Material for Safe Oral Care?

Seeking Bio-compatible Housing Material with Non-slip Grip Texture for Your Toothbrush?
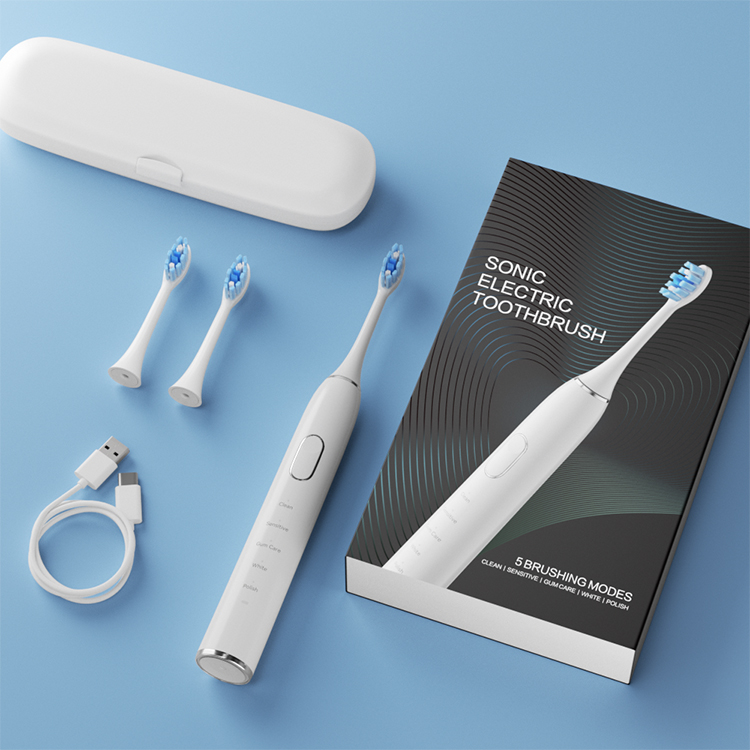
Why Is Cavitation Cleaning Technology More Effective With a Micro-Perforated Brush Head?

What Are Eco-Friendly Toothbrush Bristles Made Of? A Guide for OEM Brands