In the world of Microfluidic Chip Fabrication, precision, innovation, and flexibility are essential when developing cutting-edge technologies, especially for research and development (R&D) purposes. As the demand for microfluidic systems grows, many companies are exploring ways to streamline the prototyping and testing phases to reduce time-to-market. One key consideration for R&D teams is the Minimum Order Quantity Waiver—a service that can offer flexibility in scaling up production and testing. In this blog, we explore how Microfluidic Chip Fabrication services that offer an MOQ waiver can benefit R&D initiatives and why it’s crucial for companies looking to innovate.
Microfluidic chips are used in various applications, including diagnostics, pharmaceuticals, and environmental testing. However, the development of these chips involves multiple stages, such as design, prototyping, and testing, all of which require specialized fabrication processes. For R&D teams, the ability to experiment with small batches without committing to large production runs is invaluable. This is where Minimum Order Quantity Waiver becomes a game-changer.
By waiving the MOQ requirement, companies can test different designs, conduct experiments, and refine prototypes without the financial burden of bulk ordering. This flexibility allows researchers to optimize their designs and processes before committing to large-scale production, saving both time and money.
Prototyping in Microfluidic Chip Fabrication can often be an expensive process, especially when dealing with advanced technologies or small-batch production. Traditional manufacturers require a certain minimum quantity for orders, which may not align with the needs of R&D projects, where testing, iterating, and small runs are common.
A Minimum Order Quantity Waiver can significantly reduce initial costs by allowing R&D teams to order just the amount they need to validate their designs and test functionality. This enables the team to test concepts quickly, refine their designs, and iterate faster, which is critical for staying competitive in the fast-evolving field of microfluidics.
Innovation in microfluidics often requires rapid prototyping and testing cycles. Without the ability to start small and scale as needed, companies may face significant delays in getting their product to market. A Microfluidic Chip Fabrication service that offers an MOQ waiver makes it easier for R&D teams to develop, test, and refine prototypes in a time-efficient manner.
By eliminating MOQ barriers, R&D teams can immediately access the parts they need, perform tests, and gather data faster, speeding up the design-validation process. This quicker turnaround ultimately results in a faster time-to-market, which is especially important in competitive industries such as healthcare and diagnostics.
In R&D, flexibility and customization are often essential to ensure that prototypes meet specific project requirements. Microfluidic chips can vary significantly in terms of design, material choice, and functionality. Having access to an MOQ Waiver enables teams to request specific design modifications without committing to a large number of units.
For example, researchers might need to adjust the geometry of the microchannels or experiment with different surface treatments or materials. The flexibility to customize chip designs with smaller order quantities allows them to explore various configurations without the pressure of large-scale production runs, making it easier to optimize designs for functionality and performance.
The Microfluidic Chip Fabrication process often involves innovative designs and untested concepts that could carry a degree of risk. Without an MOQ waiver, companies might be hesitant to try out new ideas for fear of producing large quantities of faulty or unproven products. The ability to request small batches or individual prototypes minimizes this risk and allows companies to test their ideas without financial commitment.
Moreover, researchers can evaluate the chips’ performance across different use cases and environments, gathering critical data before moving forward with mass production. This lowers the likelihood of costly failures down the line and helps ensure that only the most effective designs reach the market.
Working with a Microfluidic Chip Fabrication service that offers an MOQ waiver typically also means gaining access to specialized expertise and support from the manufacturer. These companies often provide guidance on design optimization, material selection, and fabrication techniques, all of which can be invaluable during the R&D phase.
Moreover, they are often able to provide quick turnaround times, offer testing support, and help troubleshoot any issues that arise during prototyping. This additional level of support ensures that R&D teams can focus on the core challenges of their projects, knowing they have a reliable partner to guide them through the technical and operational hurdles.
For companies working on Microfluidic Chip Fabrication projects, offering an MOQ Waiver can be a strategic decision that significantly enhances the flexibility, cost-effectiveness, and speed of their R&D efforts. By reducing the barriers to prototype development, companies can test and refine their designs quickly and efficiently, improving the likelihood of success while minimizing financial risk. Whether you’re working in healthcare, environmental science, or any other field that relies on microfluidics, a Minimum Order Quantity Waiver opens the door to more rapid, cost-effective innovation. Contact us

.jpg)
whitening gel pen comparison guide: gel vs pen

What Are the Benefits of a Lithium Battery Pack in a Home Use Whitening System?

Integrating Brush Head Sterilization Technology? Need a Quiet Water Flosser Motor Too?
.jpg)
holiday photo teeth whitening: timeline for photos
.jpg)
Sonic Toothbrush Factory China

How Does Asset Tracking with RTLS Streamline Remanufacturing Core Return?
.jpg)
Water Flosser for Chick-fil-A Sauce
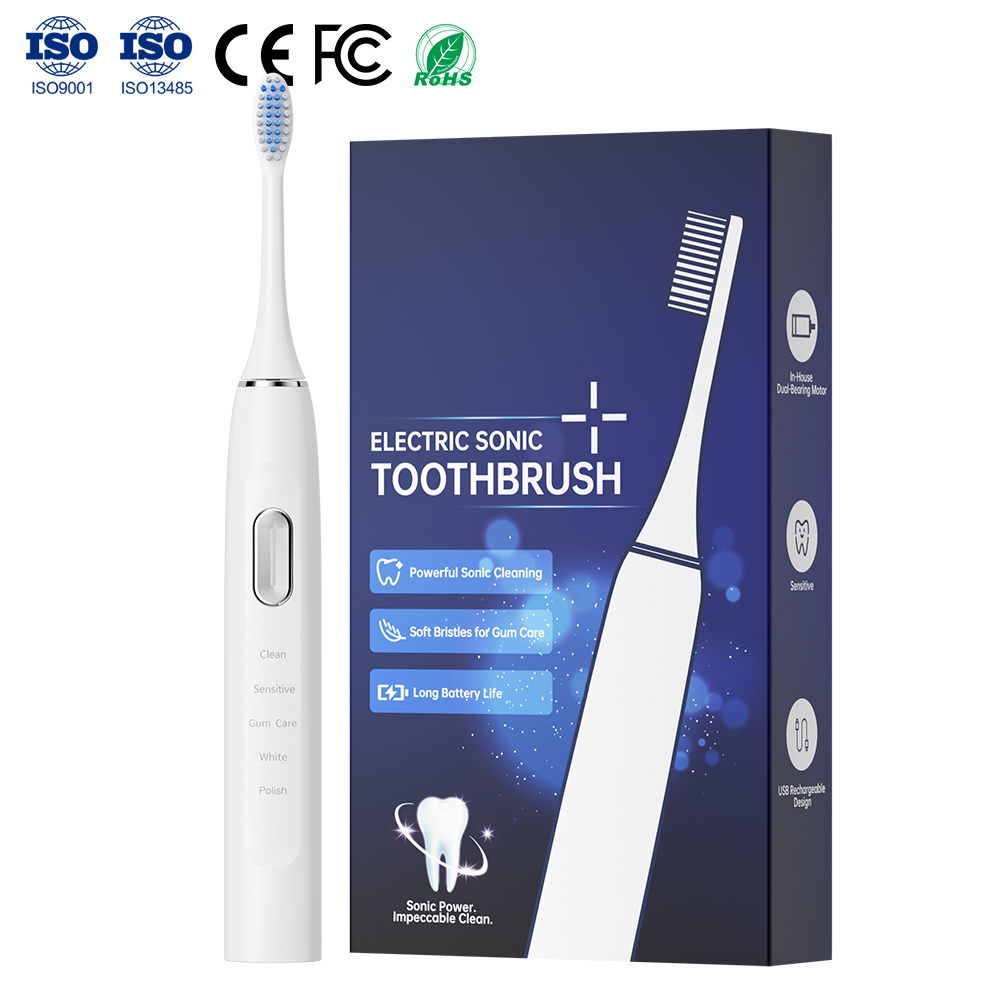
How Does Custom Handle Design Benefit from Full-scale OEM/ODM Manufacturing Capabilities?

Looking for OEM Electric Toothbrush Design and Water Flosser Private Label Services Under One Roof?
.jpg)
Maximizing Toothbrush Battery Life
.jpg)
Dry Socket Prevention Water Flosser | Safe Post-Extraction Care
.jpg)
IPX8 Electric Toothbrush Factory China
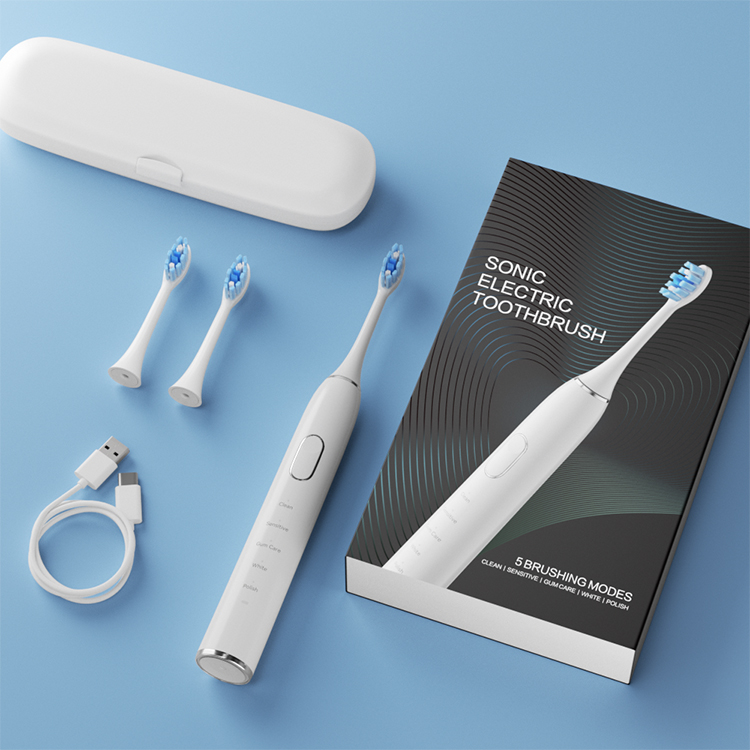
Need Packaging Customization Service for a Complete Private Label Solution?

Interested in Multi-mode Whitening Device Manufacturing and a Distributor Partnership Program Dental?

Need a Whitening Gel Filling Machine for Your Private Label Service?
.jpg)
Porcelain Veneers Whitening Guide | Maintain Veneer Brightness